 I have to adjust my company's storage tank to 1.228 or 1.216 sp.gravity at 30° c temp. Please explain what the difficulty is, rather than just starting over again. This question was asked, answered, followed up, and clarified. How should I reduced the 1.258 sp.gravity at 45c temp to 1.228 at 30c temp? Bhupesh Mulik I have a one litre of napthelene sulphonate condensate liquid of 1.258 gravity at 30c temp.I want to lower sp.gravity upto 1.228 at 30c temp by adding water.what calculation I have to do? is it depends on solid present in napthelene sulphonate condensate? Testing of concrete is a completely different field, where I doubt we can offer much help :-) Ted, you've hereby been drafted to help my daughter with her math homework next year! 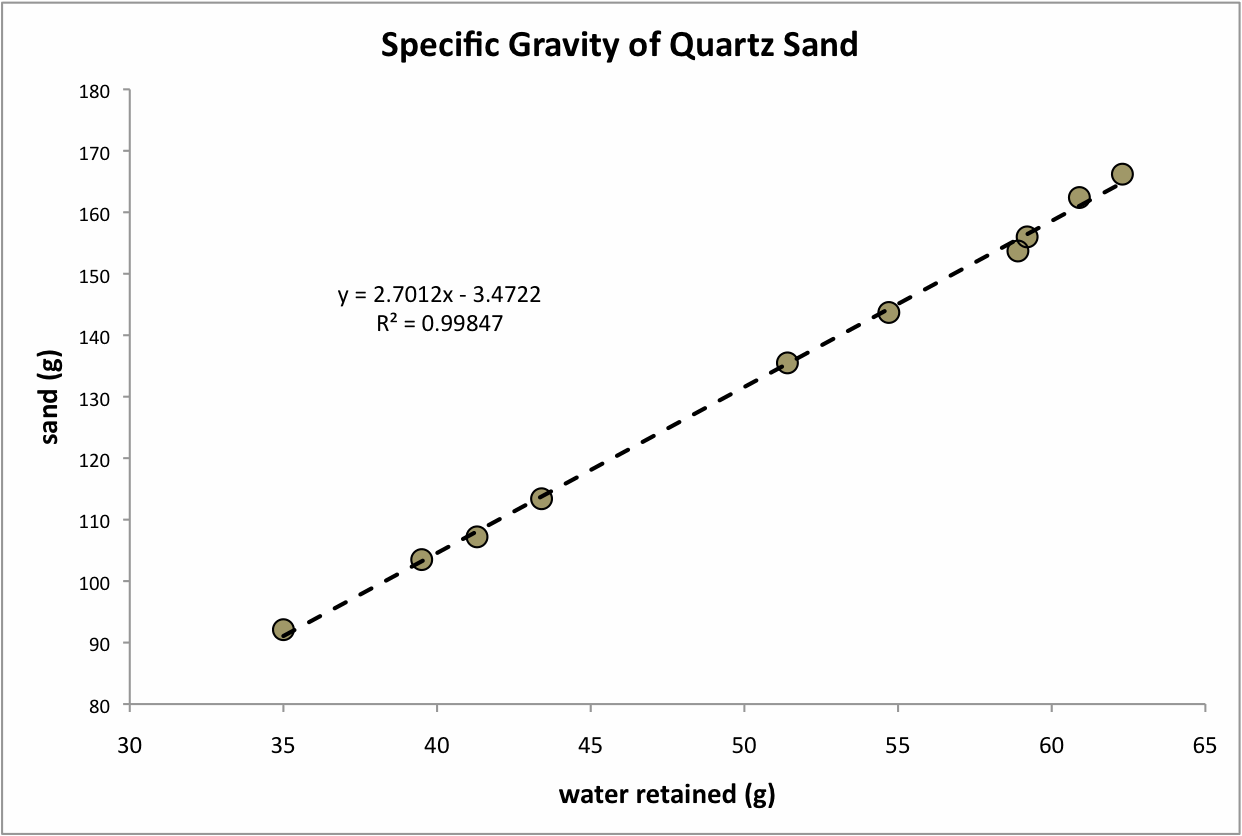
Kg/l*l + kg = (l+l) * kg/l <- the units of measure on both sides of the equation are kilograms (kg/l*kg)+(kg/l*l) = kg/l*(kg+l) <- the units of measure don't make sense So please put the units of measure into your setup of your formula,Īnd you will see that you have set it up wrong: While specific gravity is the ratio of the weight of a given volume of the substance to the weight of an equivalent volume of water, we can also say that the units of specific gravity in the metric system are kilograms per liter. My solution has 12000 kg of 1.258 sp.gravity at 30 c and I adding x amount of water to it to bring it 1.230. generally I did this type of calculation for lowering sp.gravity from 1.258 to 1.230 Those darn floaters stealing our salt).Thanks for your response. Every floater brings out both water and salt with them (at Float On we’ve crunched the numbers, and came up with 1.1 pounds of salt per floater that we lose. Another good reason to keep it slightly lower.Īt this point it may seem like you will never need to put new salt into your tank again, but alas entropy has other plans. Good for floating, but it does mean that your specific gravity will be fluctuating by minute degrees over time. We shoot to keep our specific gravity levels between 1.265 to 1.285 at all times.īe warned, the water in your tank will gradually evaporate and as you operate your tank, the specific gravity will shift higher and higher as you go. We’ve seen some float tanks with a specific gravity as low as 1.18, however within the range that works for floating people, we recommend being near the middle. The saturation point (for 93.5° F) is about 1.33. So, where should we keep the specific gravity of a float tank? A float tank will float someone starting around the 1.24 mark, and it may start to damage your pump and filtration system at or above a 1.3 specific gravity due to salt crystallization. Even molten metals have a specific gravity: lead is 11.35 and gold is 19.3. 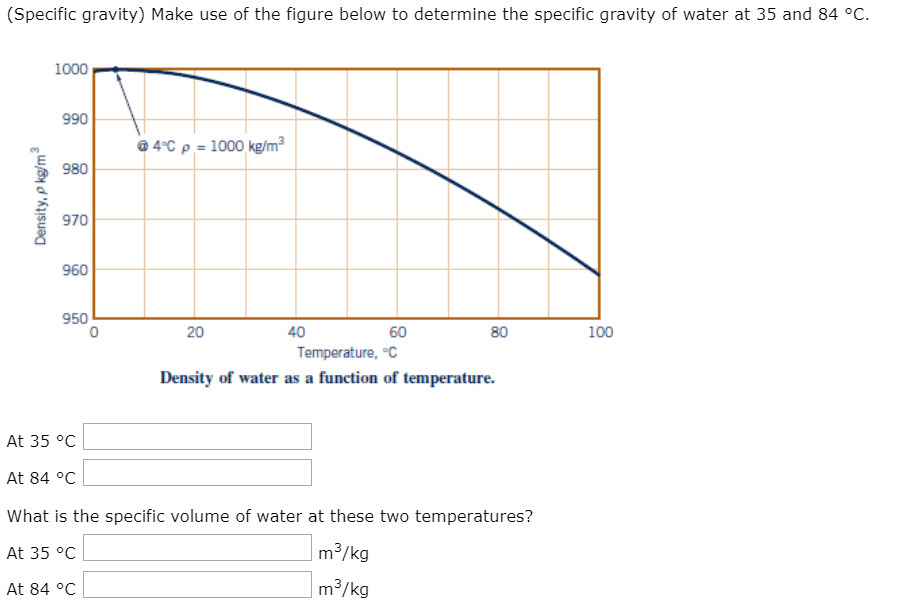
The ocean ranges from 1.023 to 1.035 and the dead sea which has been getting more saline over time is up to about 1.3 specific gravity, which is roughly where the salt reaches saturation. Water has a specific gravity of 1 (since it is being compared to itself). Specific gravity, then, in the case of our float tanks, is how dense solution is compared to regular, run of the mill water. The reference substance is nearly always water for liquids or air for gases.” “Specific gravity is the ratio of the density of a substance to the density (mass of the same unit volume) of a reference substance. After all, isn’t there just one gravity? Specific gravity refers to the gravity of a specific thing, almost always a gas or liquid. To the layman, specific gravity may seem like a misnomer. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
